Dr. Pitre's Research Fields
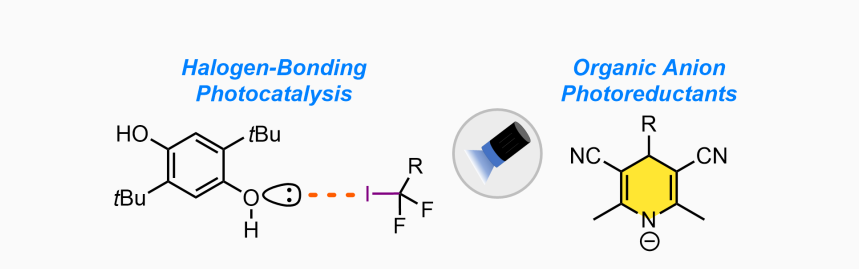
Organic Photoreductants
To further advance the field of radical chemistry, general strategies that serve to generate radicals directly from readily available precursors, such as alkyl halides, which circumvent the use of tin hydrides and ground state metal reductants would be of great value. In the Pitre Lab, we focus on developing organic single-electron photoreductants that can serve as a general platform for visible-light-mediated radical generation directly from unactivated precursors. Our research centers on two-distinct strategies: namely, halogen-bonding photocatalysis and organic anion photoreductants.
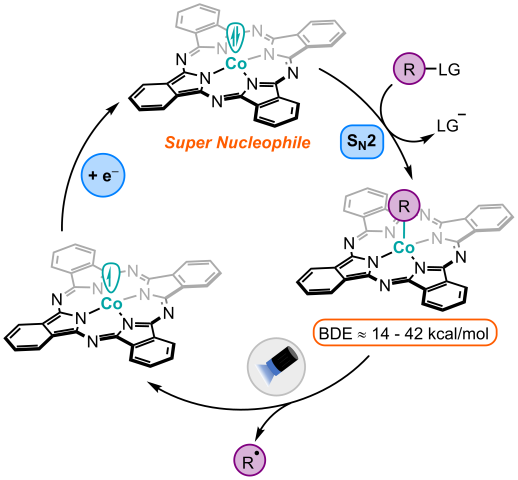
Nucleopholic Cobalt Catalysis
Macrocycle-ligated cobalt complexes, like Vitamin B12, are competent nucleophiles that undergo SN2 reactions with a variety of electrophiles, yielding an organometallic complex containing a Co–C σ-bond. A carbon-centered radical can then be generated through photolysis of this light-sensitive Co–C bond, which can often be done using visible-light irradiation from simple LED light sources. Our goal in the Pitre lab is to exploit the high nucleophilicity of cobalt macrocycles and their exceptional ability to generate carbon-centered radicals to develop novel transformations and to expand the scope of carbon-radical precursors that can be utilized in organic synthesis.